Fospropofol AHFS /Drugs.com Monograph License data
Pregnancy Dependence unknown Routes of Intravenous ATC code Legal status
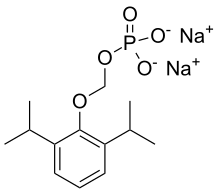
Protein binding 98%[1] Metabolism Hepatic glucuronidation Elimination half-life 0.81 hours[1] Excretion Renal show disodium [2,6-di(propan-2-yl)phenoxy]methyl phosphate
[2] CAS Number PubChem CID DrugBank ChemSpider UNII KEGG ChEMBL CompTox Dashboard (EPA ) Formula C 13 H 21 O 5 P Molar mass −1 3D model (JSmol ) show CC(C)c1cccc(c1OCOP(=O)(O)O)C(C)C
show InChI=1S/C13H21O5P/c1-9(2)11-6-5-7-12(10(3)4)13(11)17-8-18-19(14,15)16/h5-7,9-10H,8H2,1-4H3,(H2,14,15,16)
N Key:QVNNONOFASOXQV-UHFFFAOYSA-N
N N Y (what is this?)
Fospropofol (INN [3] Lusedra [4] intravenous sedative -hypnotic agent. It is currently approved for use in sedation of adult patients undergoing diagnostic or therapeutic procedures such as endoscopy .
Clinical applications [ ] Several water-soluble derivatives and prodrugs of the widely used intravenous anesthetic agent propofol have been developed, of which fospropofol has been found to be the most suitable for clinical development thus far.[5] [6] chemical compound include less pain at the site of intravenous administration, less potential for hyperlipidemia with long-term administration, and less chance for bacteremia .[citation needed Often, fospropofol is administered in conjunction with an opioid such as fentanyl.[citation needed
Clinical pharmacology [ ] Mechanism of action [ ] Fospropofol is a prodrug of propofol; it is metabolized by alkaline phosphatases to an active metabolite , propofol.
Pharmacodynamics [ ] Pharmacokinetics [ ] Initial trial results on fospropofol pharmacokinetics were retracted by the investigators. As of 2011, new results were not available.[7]
Controlled substance [ ] Fospropofol is classified as a Schedule IV controlled substance in the United States' Controlled Substances Act .[8]
References [ ]
^ Jump up to: a b Eisai Inc. (October 2009). "LUSEDRA (fospropofol disodium) Injection" (PDF) . Woodcliff Lake, New Jersey: Eisai Inc. Archived from the original (PDF) on 22 November 2010. Retrieved 2 August 2010 . ^ "Fospropofol disodium" . PubChem Compound . Bethesda, Maryland: U.S. National Library of Medicine. Retrieved 9 February 2017 .^ "Recommended INNs 2006, pt 56" (PDF) . World Health Organization . World Health Organization. Retrieved 20 April 2016 .^ "FDA Approves Fospropofol and Follows ASAs Labeling Recommendation" . American Society of Anesthesiologists. 2008-12-15. Archived from the original on 2011-05-26. Retrieved 2011-03-30 .^ Cooke A, Anderson A, Buchanan K, Byford A, Gemmell D, Hamilton N, et al. (April 2001). "Water-soluble propofol analogues with intravenous anaesthetic activity". Bioorganic & Medicinal Chemistry Letters . 11 (7): 927–30. doi :10.1016/S0960-894X(01)00088-9 . PMID 11294393 . ^ Bennett DJ, Anderson A, Buchanan K, Byford A, Cooke A, Gemmell DK, et al. (June 2003). "Novel water soluble 2,6-dimethoxyphenyl ester derivatives with intravenous anaesthetic activity". Bioorganic & Medicinal Chemistry Letters . 13 (12): 1971–5. doi :10.1016/S0960-894X(03)00346-9 . PMID 12781176 . ^ Mahajan B, Kaushal S, Mahajan R (January 2012). "Fospropofol: pharmacokinetics?" . Journal of Anaesthesiology Clinical Pharmacology . 28 (1): 134–5. doi :10.4103/0970-9185.92472 . PMC 3275955 PMID 22345970 . ^ "Schedule of Controlled Substances; Placement of Fospropofol into Schedule IV [permanent dead link
show General anesthetics (N01A )
Inhalational
Chloroethane (ethyl chloride) ‡ Chloroform ‡ Cyclopropane ‡ Desflurane Diethyl ether ‡ Enflurane Ethylene ‡ Fluroxene ‡ Halothane # Isoflurane # Methoxyflurane Methoxypropane ‡ Nitrous oxide # Sevoflurane Trichloroethylene ‡ Vinyl ether ‡ Xenon Injection
Phenols Barbiturates Opioids
Morphine # Oxycodone Anileridine ‡ Embutramide ‡ Fentanyl # Alfentanil Phenoperidine Remifentanil ÷Sufentanil Arylcyclohexylamines Neuroactive steroids Others
# WHO-EM ‡ Withdrawn from marketClinical trials :
† Phase III § Never to phase III
show Hypnotics /sedatives (N05C )
GABAA
Alcohols Barbiturates Benzodiazepines
Brotizolam Cinolazepam Climazolam Clonazepam Doxefazepam Estazolam Flunitrazepam Flurazepam Flutoprazepam Lorazepam Loprazolam Lormetazepam Midazolam Nimetazepam Nitrazepam Phenazepam Quazepam Temazepam Triazolam Carbamates Imidazoles Monoureides Neuroactive steroids Nonbenzodiazepines Phenols Piperidinediones Quinazolinones Others
GABAB
1,4-Butanediol 4-Fluorophenibut Aceburic acid Baclofen GABOB GHB (sodium oxybate )GBL GVL Phenibut Tolibut H1
Antihistamines
Captodiame Cyproheptadine Diphenhydramine Doxylamine Hydroxyzine Methapyrilene Perlapine Pheniramine Promethazine Propiomazine Antidepressants
Serotonin antagonists and reuptake inhibitors
Etoperidone Nefazodone Trazodone Tricyclic antidepressants
Amitriptyline Doxepin Trimipramine , etc.Tetracyclic antidepressants
Antipsychotics
Typical antipsychotics
Chlorpromazine Thioridazine , etc.Atypical antipsychotics
α2 -Adrenergic
Clonidine Detomidine Dexmedetomidine Lofexidine Medetomidine Romifidine Tizanidine Xylazine 5-HT2A
Antidepressants
Trazodone Tricyclic antidepressants
Amitriptyline Doxepin Trimipramine , etc.Tetracyclic antidepressants
Antipsychotics
Typical antipsychotics
Chlorpromazine Thioridazine , etc.Atypical antipsychotics
Others
Melatonin Orexin α2 δ VDCC
Gabapentin Gabapentin enacarbil Mirogabalin Phenibut Pregabalin Others
Cannabidiol
Diethylpropanediol Evoxine Fenadiazole Guaifenesin -related muscle relaxants
Chlorphenesin Mephenesin Mephenoxalone Metaxalone Methocarbamol Midaflur Opioids (e.g., morphine )Passion flower Scopolamine Trazodone UMB68 Valnoctamide
show GABA A receptor positive modulatorsAlcohols
Butanol Chloralodol Chlorobutanol (cloretone) Ethanol (alcohol) (alcoholic drink )Ethchlorvynol Isobutanol Isopropanol Menthol Methanol Methylpentynol Pentanol Petrichloral Propanol tert -Butanol (2M2P)tert -Pentanol (2M2B)Tribromoethanol Trichloroethanol Triclofos Trifluoroethanol Barbiturates Benzodiazepines Carbamates Flavonoids
Ampelopsin (dihydromyricetin) Apigenin Baicalein Baicalin Catechin EGC EGCG Hispidulin Luteolin Skullcap constituents (e.g., baicalin )Wogonin Imidazoles Kava constituents
Desmethoxyyangonin Kavain Methysticin Yangonin Monoureides Neuroactive steroids Nonbenzodiazepines Phenols Piperidinediones Pyrazolopyridines Quinazolinones Volatiles /gases
Acetone Acetophenone Acetylglycinamide chloral hydrate Aliflurane Benzene Butane Butylene Centalun Chloral Chloral betaine Chloral hydrate Chloroform Cryofluorane Desflurane Dichloralphenazone Dichloromethane Diethyl ether Enflurane Ethyl chloride Ethylene Fluroxene Gasoline Halopropane Halothane Isoflurane Kerosine Methoxyflurane Methoxypropane Nitric oxide Nitrogen Nitrous oxide Norflurane Paraldehyde Propane Propylene Roflurane Sevoflurane Synthane Teflurane Toluene Trichloroethane (methyl chloroform) Trichloroethylene Vinyl ether Others/unsorted
3-Hydroxybutanal Avermectins (e.g., ivermectin )Bromide compounds (e.g., lithium bromide , potassium bromide , sodium bromide )Carbamazepine Chloralose Chlormezanone Clomethiazole DEABL Dihydroergolines (e.g., dihydroergocryptine , , dihydroergotamine , ergoloid (dihydroergotoxine) )Efavirenz Etazepine Etifoxine Fenamates (e.g., flufenamic acid , mefenamic acid , niflumic acid , tolfenamic acid )Fluoxetine Flupirtine Hopantenic acid Lanthanum Lavender oil Lignans (e.g., 4-O-methylhonokiol , honokiol , magnolol , obovatol )Loreclezole Menthyl isovalerate (validolum) Monastrol Niacin Niacinamide Org 25,435 Phenytoin Propanidid Retigabine (ezogabine) Safranal Seproxetine Stiripentol (e.g., sulfonmethane (sulfonal) , tetronal , trional )
Terpenoids (e.g., borneol )Topiramate Valerian constituents (e.g., isovaleric acid , isovaleramide , valerenic acid , )Unsorted benzodiazepine site positive modulators: α-Pinene See also: Receptor/signaling modulators • GABA receptor modulators • GABA metabolism/transport modulators