Diazoxide
 | |
| Clinical data | |
|---|---|
| Trade names | Proglycem, Balila |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 90% |
| Metabolism | Liver oxidation and sulfate conjugation |
| Elimination half-life | 21-45 hours |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.006.063 |
| Chemical and physical data | |
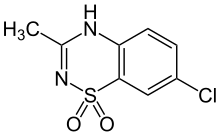
| Formula | C8H7ClN2O2S |
| Molar mass | 230.67 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 330 to 331 °C (626 to 628 °F) |
SMILES
| |
InChI
| |
Diazoxide, sold under the brand name Proglycem and Balila[1] (India), is a medication used to treat low blood sugar due to a number of specific causes.[2] This includes islet cell tumors that cannot be removed and leucine sensitivity.[2] It can also be used in refractory cases of sulfonylurea toxicity.[3] It is generally taken by mouth.[2]
Common side effects include high blood sugar, fluid retention, low blood platelets, a fast heart rate, increased hair growth, and nausea.[2] Other severe side effects include pulmonary hypertension and heart failure.[2] It is chemically similar to thiazide diuretics.[2] It works by decreasing insulin release from the pancreas and increasing glucose release by the liver.[2]
Diazoxide was approved for medical use in the United States in 1973.[2] It is on the World Health Organization's List of Essential Medicines.[4][5] It is available as a generic medication.[6]
Medical uses[]
Diazoxide is used as a vasodilator in the treatment of acute hypertension or malignant hypertension.[7]
Diazoxide also inhibits the secretion of insulin by opening ATP-sensitive potassium channel of beta cells of the pancreas; thus, it is used to counter hypoglycemia in disease states such as insulinoma (a tumor producing insulin)[8] or congenital hyperinsulinism.
Diazoxide acts as a positive allosteric modulator of the AMPA and kainate receptors, suggesting potential application as a cognitive enhancer.[9]
Side effects[]
This article needs additional citations for verification. (July 2015) |
Diazoxide interferes with insulin release through its action on potassium channels.[10] Diazoxide is one of the most potent openers of the K+ ATP channels present on the insulin producing beta cells of the pancreas. Opening these channels leads to hyperpolarization of cell membrane, a decrease in calcium influx, and a subsequently reduced release of insulin.[3] This mechanism of action is the mirror opposite of that of sulfonylureas, a class of medications used to increase insulin release in Type 2 Diabetics. Therefore, this medicine is not given to non-insulin dependent diabetic patients.
The Food and Drug Administration published a Safety Announcement in July 2015 highlighting the potential for development of pulmonary hypertension in newborns and infants treated with this drug.[11]
See also[]
References[]
- ^ "Diazoxide Capsule". indiamart.com. Archived from the original on 2021-02-21. Retrieved 2021-02-06.
- ^ a b c d e f g h "Diazoxide Monograph for Professionals". Drugs.com. Retrieved 11 October 2019.
- ^ a b Doyle, Máire E.; Egan, Josephine M. (2003-03-01). "Pharmacological Agents That Directly Modulate Insulin Secretion". Pharmacological Reviews. 55 (1): 105–131. doi:10.1124/pr.55.1.7. ISSN 1521-0081. PMID 12615955. S2CID 11121340.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 708. ISBN 9780857113382.
- ^ van Hamersvelt HW, Kloke HJ, de Jong DJ, Koene RA, Huysmans FT (August 1996). "Oedema formation with the vasodilators nifedipine and diazoxide: direct local effect or sodium retention?". Journal of Hypertension. 14 (8): 1041–5. doi:10.1097/00004872-199608000-00016. PMID 8884561. S2CID 3283469.

- ^ Huang Q, Bu S, Yu Y, et al. (January 2007). "Diazoxide prevents diabetes through inhibiting pancreatic beta-cells from apoptosis via Bcl-2/Bax rate and p38-beta mitogen-activated protein kinase". Endocrinology. 148 (1): 81–91. doi:10.1210/en.2006-0738. PMID 17053028.

- ^ Randle, John C.R.; Biton, Catherine; Lepagnol, Jean M. (15 November 1993). "Allosteric potentiation by diazoxide of AMPA receptor currents and synaptic potentials". European Journal of Pharmacology. 247 (3): 257–65. doi:10.1016/0922-4106(93)90193-D. PMID 8307099.

- ^ Panten, Uwe; Burgfeld, Johanna; Goerke, Frank; Rennicke, Michael; Schwanstecher, Mathias; Wallasch, Andreas; Zünkler, Bernd J.; Lenzen, Sigurd (1989-04-15). "Control of insulin secretion by sulfonylureas, meglitinide and diazoxide in relation to their binding to the sulfonylurea receptor in pancreatic islets". Biochemical Pharmacology. 38 (8): 1217–1229. doi:10.1016/0006-2952(89)90327-4. PMID 2650685.
- ^ "FDA Drug Safety Communication: FDA warns about a serious lung condition in infants and newborns treated with Proglycem (diazoxide)" (Press release). Food and Drug Administration. July 16, 2015. Retrieved 2015-07-19.
External links[]
- "Diazoxide". Drug Information Portal. U.S. National Library of Medicine.
- Carbonic anhydrase inhibitors
- Vasodilators
- Potassium channel openers
- AMPA receptor positive allosteric modulators
- Kainate receptor agonists
- Benzothiadiazines
- Sultams
- Chloroarenes
- World Health Organization essential medicines